Giovanna Forte is a PR and communications specialist who became Managing Director of Forte Medical after she joined forces with her brother, Dr. Vincent Forte, who had designed a medical device that set out to save millions of pounds for the NHS. The pair developed the product together and in 2006 she secured investment funding for the company, which designs specimen collection systems that make right-first-time analysis, diagnosis and treatment implicit to routine medicine. Today the company is a supplier to the NHS and exports to the U.S.

“…As soon as the U.S. shows steady growth, I will turn my attention to wider export markets; Europe, Australasia and the Far East are calling…”
Giovanna, please can you tell us about your career to date and what made you move into the medical sector?
I started life as a PA, worked for high-level bosses, landing up in the world of trade exhibitions. Aged 23, I met my husband-to-be. By age 26 I had our first daughter, an event that coincided with the 1990s recession. My plans to be a lady-at-lunch never materialised; I had to get a job. Leaving my one month old child didn’t appeal, so I picked up the phone to all my exhibition contacts and asked if they would give me work as a support services consultant. They said yes, and Fortissima! (which went on to become Forte Communication) was born.
By the time my second daughter arrived, the business had flourished; within a few more years, Ab-Fab days were upon me with an office in Soho. Forte Communication did very well with clients from architecture, engineering, education and brands.

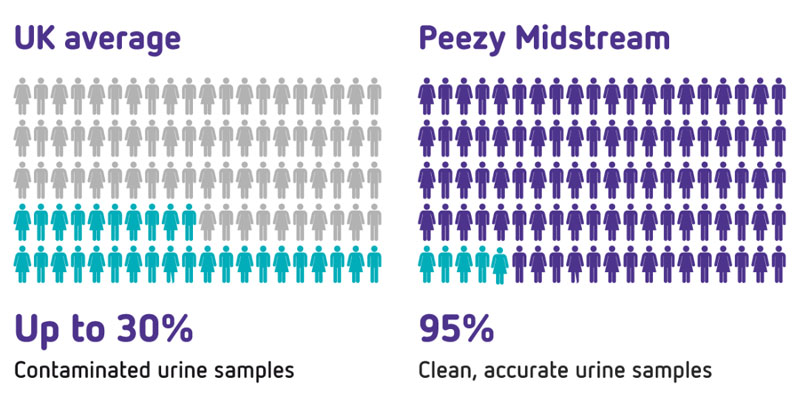
Meanwhile in Suffolk, my NHS GP brother Dr. Vincent Forte had been struggling to diagnose and treat women patients who presented with urinary tract infections (UTIs). Being an inquisitive sort he found that a high proportion of urine specimens were contaminated before they reached the laboratory. This meant that the lab may as well be looking through a dirty window; results were often inconclusive and he had to rely on broad spectrum antibiotics that did not always work.
This type of mixed growth contamination is largely due to natural bacteria being washed off the skin into the specimen but it renders results inconclusive. Once he understood this, he invented a concept that would reject the first-stream, capture and isolate the midstream urine (MSU) and get rid of any excess urine into the loo.
The Female Freedom Funnel was born and promptly won a Medical Futures Award. Vincent asked me to help take his innovation to market. For five years we worked off-and-on, he maintaining his GP job and me working hard with the PR agency.
We funded things personally over this time, but by 2006 ran out of ready cash and decided to seek external funding. By now our product was renamed Peezy Midstream and prototyped. We secured investment through a Government Early Growth scheme with matched angel funding, most of which came through friends and family.
Government of course wanted the company to have a full time leader. I closed Forte Communication to focus on Forte Medical. Although we remained co-directors, Vincent continued his work as a GP, while I built our business. In 2012 we started to sell to the NHS and private sector in the UK; growing our export markets is a more recent, very rewarding string to our bow.
Please can you tell us what your role at Forte Medical involves on a day to day basis?
Everything! From 2006 to 2013 I worked alone on product development, patents, quality assurance, manufacture, fundraising, marketing and sales. It was tough. By 2013 I took in more funding and could afford a manager who is still with me today. I now focus on UK and export sales, marketing, lobbying and manufacture. I have also set the scene for company growth with a portfolio of new products.
You currently export to the U.S. – please can you tell us how this came about?
A shareholder in the healthcare business found himself next to a U.S. diagnostics company at an exhibition. Being a joiner-of-dots, he told his U.S. neighbour about us, forged the introduction and the rest has evolved over the last twelve months with collaborative research, evaluations and negotiation.
Happily the introduction coincided with the start of a clinical trial at Stanford Medical School too. Our partner is now our lead distributor for the U.S.; he in turn is appointing regional distributors some of which have already signed up to significant volume sales.
You’ve created branded products – how did you go about protecting your intellectual property at home and overseas?
We were lucky enough to have a brilliant IP [Intellectual Property] lawyer and highly diligent patent attorney from day one (the details are on our website). We spent personal funds on patent applications until external investment took over. We have granted patents in all major territories where we might sell or manufacture. This is an expensive but absolutely essential aspect of any business involved in innovation.
What are the greatest challenges you’ve faced and how have you overcome them?
Challenge one: Pee is not sexy! “It’s only urine” is an oft-heard throwaway from clinicians and nurses the world over. If blood samples were treated with the same cursory attitude, there would be an outcry. Urine is a specimen from which people are diagnosed. From the 65 million urine specimens collected annually by the NHS, mixed growth rates can be as high as 70%. This means millions of pounds are being flushed down hospital loos daily all over the world. We intend to bring universal diligence to this overlooked area of medicine.
Challenge two: NHS privatisation. Private screening and laboratory services to whom the NHS outsource are often paid on volume. Reducing retests will reduce profit. It’s a ridiculous anomaly in compensation structure. Someone didn’t think things through and its costing our health service – and its patients – dear.
Challenge three: A few years ago when fundraising outside of the friends and family circle I encountered “businessmen” who tried to steal the company from under our feet. Of course, if other people want what we have, it means it is worth having, but it has sometimes been incredibly stressful.
What sort of support have you had from UKTI?

UKTI [UK Trade & Investment] has engaged with us on a number of occasions. The problem is that our business is very niche; specimen collection isn’t the most widely analysed or understood sector within healthcare, which means that often the market intelligence and research data needed for market access simply isn’t there. Once we are established in a variety of export markets, UKTI will be better placed to assist with business on the ground.
What is your advice for women looking to export?
Belief in your product or service is essential. Then find out if what you have to offer is relevant to export markets, which ones and how. Carry out really thorough market research, attend appropriate events, find local advocates, establish evidence and data and go for it. A local sales partner is usually a good idea. I’m a firm believer that if you really want to do something – anything – you can do it. Think logically, carefully and strategically and you’ll win through. Oh, and don’t try and do too much in one go.
What is coming up next for you and Forte Medical?
As soon as the U.S. shows steady growth, I will turn my attention to wider export markets; Europe, Australasia and the Far East are calling. My ambition is for urine analysis to have a global gold standard that relies on the Peezy brand and products.
The company’s future lies in The Specimen Collection, a portfolio of Peezy brand products that deliver accurate specimen collection for early stage prostate and cervical cancer urine tests. Semen and stool are also on our radar … which means an already woeful number of dinner party invitations will simply evaporate!
https://twitter.com/Forte_Medical
https://www.facebook.com/PeezyMidstream
https://www.linkedin.com/in/giovannaforte